California Patent of the Month – August 2023
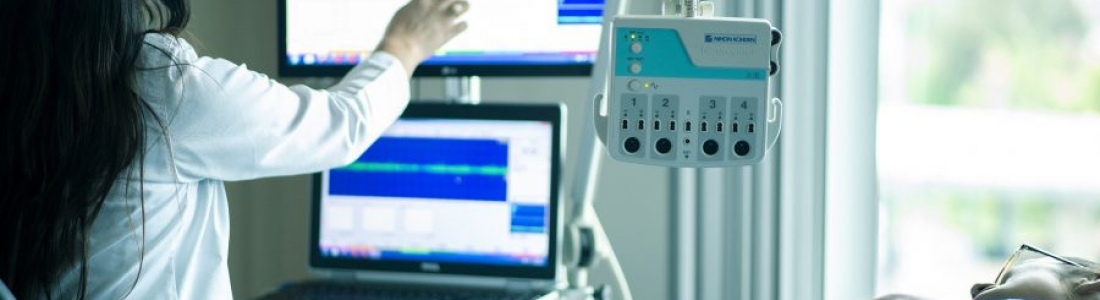
Innovations in medical technology continue to revolutionize the field of healthcare, offering new possibilities for treating neurological disorders and improving patient outcomes. One such groundbreaking invention is the implantable medical device developed by NeuroPace, Inc., a leader in neurostimulation therapies. NeuroPace’s RNS® System is the only FDA-approved epilepsy device that delivers personalized treatment by responding to abnormal brain activity and provides EEG data that can help improve patient care.
The newly patented multimodal brain sensing device combines advanced sensing modalities with precise timing schedules to acquire continuous measurements related to brain activity, enabling targeted and responsive treatment for a range of conditions.
The implantable medical device features a lead with macroelectrodes, microelectrodes, and optical sensors. These components enable the implementation of multiple sensing modalities, providing a comprehensive understanding of brain activity.
A host device is connected to the lead and operates according to a timing schedule that facilitates time multiplexed continuous measurement acquisition. This means that different sensing modalities are implemented at specific intervals, ensuring diverse measurements while allowing each modality to pause when others are implemented. By sharing the same type of lead component, the host device controls and coordinates the implementation of at least two different sensing modalities.
The macroelectrodes are strategically spaced along the length of the lead body, while the microelectrodes are positioned around the circumference of the lead’s distal portion. Additionally, the optical sensor assemblies are placed between pairs of macroelectrodes. This arrangement optimizes the coverage and accuracy of the measurements obtained from various brain regions.
The different sensing modalities encompass a wide range of measurements. Examples include field potential and rheoencephalography measurements implemented by the macroelectrodes, single unit activity and multi-unit activity measurements conducted by the microelectrodes, and optical blood volume or optical blood oxygenation measurements facilitated by the optical sensor assemblies.
Notably, the host device can output electrical stimulation signals to the macroelectrodes while suspending certain sensing modalities. This feature allows for effective integration of stimulation therapy with measurement acquisition, enhancing the overall treatment efficacy.
The implantable medical device system developed by NeuroPace represents a significant advancement in the field of neurostimulation and responsive therapies. NeuroPace’s commitment to innovation and their focus on improving patient outcomes position them at the forefront of neurostimulation therapies, bringing hope and relief to individuals living with neurological disorders worldwide.
Are you developing new technology for an existing application? Did you know your development work could be eligible for the R&D Tax Credit and you can receive up to 14% back on your expenses? Even if your development isn’t successful your work may still qualify for R&D credits (i.e. you don’t need to have a patent to qualify). To find out more, please contact a Swanson Reed R&D Specialist today or check out our free online eligibility test.
Who We Are:
Swanson Reed is one of the U.S.’ largest Specialist R&D tax advisory firms. We manage all facets of the R&D tax credit program, from claim preparation and audit compliance to claim disputes.
Swanson Reed regularly hosts free webinars and provides free IRS CE and CPE credits for CPAs. For more information please visit us at www.swansonreed.com/webinars or contact your usual Swanson Reed representative.