Quick Answer: Wisconsin R&D Tax Credit Framework This study provides an exhaustive analysis of the United States federal and Wisconsin state Research and Development (R&D) tax credit requirements. It details the statutory frameworks, including the federal Four-Part Test and Wisconsin’s localized 5.75% and 11.5% enhanced credit tiers, and applies them directly to five vital industries within the Madison, Wisconsin innovation ecosystem: Biotechnology, Healthcare IT, Medical Imaging, Nuclear Engineering, and Agricultural Technology.
This study provides an exhaustive analysis of United States federal and Wisconsin state Research and Development tax credit requirements, applying these statutory frameworks to five distinct industries within the Madison, Wisconsin innovation ecosystem. Through detailed historical context, legislative guidance, and judicial precedent, this document outlines the eligibility criteria and compliance strategies for localized enterprises.
The Statutory Framework of the Research and Development Tax Credit
The Research and Development (R&D) Tax Credit is a foundational fiscal incentive designed to stimulate domestic investment in technological innovation, process improvement, and the development of new business components. For enterprises operating in Madison, Wisconsin, understanding the parallel but distinct frameworks of the United States federal government and the State of Wisconsin is critical for maintaining tax compliance and maximizing available capital. The landscape of research taxation is governed by a complex intersection of statutory law, administrative directives from tax authorities, and a continuously evolving body of judicial precedent.
The United States Federal R&D Tax Credit (IRC Section 41 and 174)
The federal R&D tax credit, codified under Title 26 of the United States Code (U.S.C.) Section 41, provides a dollar-for-dollar reduction in federal income tax liability for qualified research expenses (QREs). First enacted in 1981, the credit was explicitly designed to reward businesses that undertake severe technical challenges and incur the financial risks inherently associated with domestic innovation. The legislative intent is to prevent the erosion of the United States’ technological superiority by subsidizing the high costs of research personnel, experimental supplies, and contracted research services.
Concurrently, Internal Revenue Code (IRC) Section 174 dictates the specific accounting and tax treatment of research and experimental (R&E) expenditures. Historically, taxpayers were permitted to immediately expense these developmental costs in the year they were incurred, providing an immediate cash flow benefit. However, the regulatory environment experienced a massive paradigm shift with the passage of the Tax Cuts and Jobs Act (TCJA) of 2017. Under the TCJA, for taxable years beginning after December 31, 2021, taxpayers were mandated to capitalize and amortize domestic R&E expenditures over a five-year period, and foreign research over a fifteen-year period. This amortization requirement severely impacted the cash flow of innovation-heavy industries, forcing companies to defer the tax benefits of their research investments over half a decade.
Recently, the legislative landscape shifted again with the enactment of Public Law 119-21, commonly known as the One Big Beautiful Bill Act (OBBBA). The OBBBA introduced a new provision, Section 174A, which permanently restored the ability of taxpayers to fully expense domestic research or experimental expenditures paid or incurred in taxable years beginning after December 31, 2024. Furthermore, the legislation provided critical transition options under Section 70302(f), allowing taxpayers to recover unamortized amounts paid or incurred during the 2022 through 2024 gap years. As clarified by IRS Revenue Procedure 2025-28, taxpayers must navigate specific procedural mechanisms to implement these transition rules and begin applying either Section 174A(a) for immediate expensing or Section 174A(c) for an elective 60-month amortization of capital accounts. Despite this relief for domestic activities, the OBBBA maintained the strict fifteen-year capitalization requirement for foreign research, thereby heavily incentivizing companies to localize their R&D operations within the United States.
The Federal Four-Part Test
To be eligible for the federal credit under Section 41, a taxpayer must demonstrate that their activities meet the rigorous, statutory “Four-Part Test” outlined in the Internal Revenue Code and supporting Treasury Regulations. The Internal Revenue Service (IRS) mandates that every individual business component—whether a product, process, software, technique, formula, or invention—evaluated for the credit must independently satisfy all four of the following criteria. Failure to satisfy even a single prong of this test results in the complete disallowance of the associated qualified research expenses.
| Test Component |
Statutory Requirement |
Practical Application and Nuance |
| Permitted Purpose |
The activity must relate to developing or improving the functionality, quality, reliability, or performance of a business component. |
Aesthetic, cosmetic, or seasonal design changes are explicitly excluded from eligibility. The taxpayer’s objective must be rooted in functional or performance-based enhancements. |
| Technological in Nature |
The development process must fundamentally rely on the established principles of the hard sciences, specifically physical science, biological science, engineering, or computer science. |
Research based in economics, humanities, arts, social sciences, or general business management is strictly prohibited. The courts require personnel to possess the requisite scientific background to conduct such activities. |
| Elimination of Uncertainty |
At the outset of the developmental project, the taxpayer must encounter genuine technical uncertainty regarding the capability, method, or appropriate design of the business component. |
The taxpayer is not required to guarantee ultimate commercial success. Discovering that a particular scientific method fails is considered a valid resolution of uncertainty under the tax code. |
| Process of Experimentation |
Substantially all activities must constitute elements of a process of experimentation, which includes testing, modeling, simulating, or systematic trial and error. |
This requires a methodical, documented plan: forming a scientific hypothesis, testing alternatives, analyzing quantitative data, and refining the approach based on those results. |
In addition to the Four-Part Test, taxpayers must navigate the complexities of IRC Section 280C(c). Taxpayers may elect a reduced R&D credit to avoid reducing their corresponding Section 174 deduction by the amount of the credit claimed. This election, made on IRS Form 6765, requires careful financial modeling to determine the optimal tax posture for the enterprise.
The Wisconsin State R&D Tax Credit Framework
The Wisconsin Department of Revenue (DOR) administers a robust state-level R&D credit that largely conforms to the federal definitions of qualified research under IRC Section 41, but introduces several critical statutory deviations, enhanced benefits, and distinct reporting requirements designed to aggressively promote localized economic growth. Wisconsin law, codified under Chapter 71 of the Wisconsin Statutes, explicitly requires that eligible research must be conducted strictly within the state’s geographic borders.
A paramount distinction between the federal and state frameworks is Wisconsin’s approach to the recent federal accounting changes. Wisconsin has explicitly decoupled from the federal Tax Cuts and Jobs Act modifications regarding IRC Section 174. For state corporate income and franchise tax purposes, Wisconsin references the Internal Revenue Code in effect on December 31, 2021. Consequently, Wisconsin continues to allow the immediate, full expensing of research and experimental costs for activities conducted within the state, completely bypassing the complex five-year federal amortization rules that burdened taxpayers between 2022 and 2024.
Wisconsin offers a highly nuanced, tiered credit rate system based on the specific technological nature of the research being performed:
| Credit Tier |
Statutory Rate |
Eligibility Criteria |
| General Research Credit |
5.75% |
The credit equals 5.75% of the amount by which Wisconsin QREs for the taxable year exceed 50% of the average QREs for the three preceding taxable years. For startups with no prior QREs, the rate is 2.875% of the current year’s expenses. |
| Enhanced Credit: Engines |
11.5% |
Under Wis. Stat. § 71.28(4)(ab)2, research related to designing internal combustion engines qualifies for an 11.5% rate on excess QREs. Statutorily, this explicitly includes substitute products such as fuel cell, electric, and hybrid drives. |
| Enhanced Credit: Energy Efficiency |
11.5% |
Research activities directed toward certain energy-efficient products—such as advanced lighting systems, building automation and control systems, or automotive batteries for hybrid vehicles—qualify for the 11.5% rate. |
Furthermore, Wisconsin has recognized that early-stage technology companies often operate at a net loss and cannot immediately utilize nonrefundable tax credits. To address this, the legislature introduced a lucrative refundability mechanism. For taxable years beginning between January 1, 2021, and January 1, 2024, up to 15% of the research credit was refundable. For taxable years beginning on or after January 1, 2024, the refundable portion significantly increased to 25%. This allows pre-revenue startups to receive an immediate cash injection from the Department of Revenue, while any unused nonrefundable portions may be carried forward for up to fifteen years to offset future tax liabilities. Pass-through entities, such as S-Corporations and Partnerships, allocate these credits pro-rata to their owners via Schedule K-1, reporting the activity on Wisconsin Schedule R.
In addition to income and franchise tax credits, Wisconsin provides a highly advantageous sales and use tax exemption. This exemption applies to the purchase of machinery, equipment, parts, accessories, and tangible personal property (including fuel and electricity) that is consumed or destroyed while being used exclusively and directly in qualified research. This exemption is specifically targeted at persons engaged in manufacturing or biotechnology within the state, providing a profound reduction in capital expenditure overhead for hardware-intensive research facilities.
The Madison, Wisconsin Innovation Ecosystem
Madison, Wisconsin, the state capital and a geographic nexus of academic and commercial enterprise, has evolved over the past century into a premier geographic cluster for research and development. This sustained economic transformation is deeply rooted in the institutional infrastructure provided by the University of Wisconsin-Madison (UW-Madison) and its historically powerful technology transfer arm, the Wisconsin Alumni Research Foundation (WARF).
Investing approximately $1.9 billion annually in diverse scientific research, UW-Madison consistently ranks fifth in the nation for total research spending and serves as the fundamental economic engine for Dane County and the broader state. The resulting innovation ecosystem generates an estimated $30.8 billion annual statewide economic impact, an economic footprint equivalent to nearly nine percent of Wisconsin’s total economy. This academic-industrial complex is responsible for supporting over 232,000 jobs across the public and private sectors, generating $1 billion in state and local taxes, and delivering a massive return on investment for taxpayers. A study by NorthStar Analytics indicates that for every single dollar invested by taxpayers into the university, more than $26 is injected back into the state’s economy.
WARF is the institutional glue that binds academic discovery to commercial realization. Founded in 1925 by UW-Madison biochemist Harry Steenbock to protect his groundbreaking vitamin D discoveries, WARF was one of the first university technology transfer offices in the world. Operating with a mission to promote, encourage, and aid scientific investigation, WARF has managed the intellectual property for over 3,000 UW-Madison discoveries. By licensing these technologies to private industry, WARF generates massive royalty streams, from which it consistently plows $25 million to $30 million back into the UW-Madison Graduate School each year to support foundational basic research.
This cyclical reinvestment model has spawned over 400 startups directly from university research and attracted major multinational corporations to establish specialized, large-scale R&D facilities within the Madison area, particularly in the University Research Park. The Wisconsin Economic Development Corporation (WEDC) actively supports this cluster, identifying biohealth, energy, advanced manufacturing, and water technology as key industries for strategic state investment. The geographic concentration of specialized talent—driven by the university’s production of over 750 PhDs annually—has allowed distinct industry verticals to mature. Recent labor market analytics consistently rank Madison in the top 20 United States markets for life sciences research and development, biomedical engineering, and advanced medical technology manufacturing, possessing the highest concentration of microbiologists, chemists, and biological technicians among the top 100 markets.
The subsequent sections of this study provide an exhaustive analysis of five unique industry case studies. These case studies detail the historical evolution of specific sectors within Madison, demonstrating precisely how localized entities navigate the complex technological requirements, regulatory hurdles, and strict legal precedents necessary to successfully claim the United States federal and Wisconsin state R&D tax credits.
Case Study 1: Biotechnology and Life Sciences
Industry Evolution in Madison
The biotechnology and life sciences sector in Madison is the direct, linear descendant of a century of pioneering biochemical research conducted at UW-Madison. The industry’s foundational moment occurred in the 1920s when Professor Harry Steenbock discovered a revolutionary method to increase the Vitamin D content of foods via ultraviolet irradiation, a breakthrough that ultimately led to the worldwide eradication of rickets. Recognizing the immense commercial potential and vital public health value of this discovery, Steenbock resisted the urge to sell the patent to a single corporation, instead partnering with alumni to establish WARF in 1925 to license the technology broadly. This established a precedent of patenting and commercializing biological discoveries that has defined Madison for a century.
During the early 1990s, the local industry underwent a paradigm shift. Historically, WARF had exclusively licensed patents to massive, established external pharmaceutical corporations. However, under the leadership of Managing Director Dick Leazer, WARF shifted its strategy to actively invest equity in untested university startups. This pivot catalyzed the local industry, exemplified by the 1993 founding of Third Wave Technologies. Third Wave was born from a fortuitous collaboration between UW-Madison chemistry professor Lloyd Smith (who had invented automated DNA sequencing methods) and biomolecular chemistry professor James Dahlberg (who identified enzymes capable of detecting human genetic mutations). WARF’s decision to support this fledgling company with startup equity investment ignited a wave of localized biotech entrepreneurship.
Recognizing an acute need for a specialized workforce capable of bridging the difficult gap between bench science and commercial product launch, the UW-Madison School of Medicine and Public Health, heavily backed by the state administration of Governor Tommy Thompson, launched the Master of Science in Biotechnology program in 2002. This unique, cross-functional curriculum fused hard science, business strategy, regulatory policy, and intellectual property law. Producing over 400 graduates—the vast majority of whom remained in Wisconsin—the program supplied the critical human capital required to scale local startups into global entities. Today, Madison boasts a robust biohealth cluster comprising over 2,000 establishments, including massive global entities like Promega, Exact Sciences, Illumina, and Aldevron, all of which operate highly sophisticated research and biomanufacturing facilities fueled by localized intellectual capital.
R&D Tax Credit Application and Eligibility
Biotechnology firms in Madison, ranging from highly leveraged early-stage oncology startups to massive biomanufacturing entities, consistently engage in experimental activities that meet both the federal Four-Part Test and the Wisconsin state criteria. Because their core operations revolve around manipulating biological systems, establishing the “Technological in Nature” requirement is typically straightforward, provided the proper documentation is maintained.
Qualifying Activities in Biotechnology:
- Formulation and Assay Development: Companies attempting to discover novel genetic markers, optimize reagents for molecular diagnostics, or develop proprietary compounds incur massive wage and supply expenses. Analytical scientists, discovery biology specialists, and formulation chemists engaged in iterative testing of these compounds qualify for the credit.
- Clinical Trials: The design, execution, and rigorous analytical review of clinical trial data by pharmacology associates, clinical trial managers, and biometrics associates to establish the safety, toxicity, and efficacy of a new biologic or pharmaceutical intervention.
- Process Engineering and Scale-Up: Translating the manufacturing of a complex biological compound from small, laboratory-scale yields to massive commercial biomanufacturing yields. This involves overcoming severe technical uncertainties related to fluid dynamics, thermodynamics, mechanical shear forces, and the prevention of biological contamination within large bioreactors.
Case Law and Administrative Guidance: When claiming credits for biotechnology, firms must remain highly vigilant regarding the strict legal application of the “Technological in Nature” test. As observed in broader biological and agricultural cases such as the recent Tax Court decision in George v. Commissioner, the court emphasizes that the research must be firmly rooted in the hard sciences rather than general operational optimization or standard animal husbandry. In George, the court disallowed a significant portion of the taxpayer’s claims because they lacked personnel capable of conducting true scientific research, emphasizing that merely operating in a science-adjacent field does not automatically qualify daily operations as experimentation. Madison biotech firms must ensure that the personnel whose wages are claimed possess the requisite scientific degrees or technical backgrounds to conduct valid hypothesis-driven research.
Furthermore, a defining characteristic of the modern biotechnology industry is the heavy reliance on Contract Research Organizations (CROs). Under IRC Section 41, expenses paid to third-party contractors to conduct research on the taxpayer’s behalf generally qualify at 65% of the total invoiced cost (or 75% if paid to a qualified research consortium). However, the taxpayer must carefully navigate the “Funded Research Exclusion”.
The seminal case Fairchild Industries, Inc. v. United States established the framework for this exclusion. The Federal Circuit Court of Appeals ruled that for third-party or contracted research to be eligible, the taxpayer must bear the financial risk of failure. If a Madison-based biotech firm executes a fixed-fee contract where payment is strictly contingent on the CRO successfully delivering a viable, functional molecule, the CRO—not the Madison firm—is viewed as bearing the economic risk, thus altering who is legally entitled to the R&D credit. Conversely, if the biotech firm pays the CRO on a “time-and-materials” basis, paying for the labor regardless of the ultimate scientific outcome, the Madison firm successfully establishes financial risk and may claim the expenses. Wisconsin strictly mirrors this framework, requiring meticulous contract review by the Department of Revenue to ensure the activity is not statutorily disqualified as funded research.
Case Study 2: Healthcare Information Technology (The Epic Systems Cluster)
Industry Evolution in Madison
Madison is globally recognized as an undisputed epicenter for healthcare information technology (IT) and electronic health records (EHR). The gravitational anchor of this massive technological cluster is Epic Systems Corporation, located on a sprawling, whimsical 1,100-acre campus in the adjacent suburb of Verona, Wisconsin. The history of Epic is a testament to the power of localized software engineering talent.
Founded in 1979 by Judy Faulkner, who holds a master’s degree in computer science from UW-Madison, the company originally started under the name Human Services Computing. Operating out of a basement office in a Madison apartment building with a single Data General Eclipse computer and an initial investment of $70,000 from friends and family, Faulkner set out to address severe inefficiencies in healthcare data management. Initially focused on database management systems for scheduling appointments and billing, Epic recognized early the monumental paradigm shift toward comprehensive clinical documentation.
The release of EpicCare in 1992, a pioneering Windows-based electronic medical record system, revolutionized the healthcare IT industry. Epic consistently reinvested its revenue into aggressive software development, launching the MyChart patient portal in 2000, which fundamentally altered patient engagement. By 2003, Epic secured a massive $1.8 billion contract with Kaiser Permanente to digitize records for over 8 million patients, cementing its status as an industry titan. When the federal HITECH Act of 2009 injected $27 billion into the U.S. healthcare system to incentivize electronic record adoption, Epic was perfectly positioned for exponential, aggressive growth.
Today, Epic Systems generates over $4.6 billion in annual revenue, employs roughly 13,000 people, and maintains the medical records for over 305 million patients globally. Epic’s decision to remain a privately held company, devoid of public market pressures, has allowed it to maintain a relentless focus on long-term software architecture rather than quarterly earnings. The massive, localized presence of Epic has spawned a dense ecosystem of specialized IT consulting firms, bespoke software developers, and third-party healthcare analytics vendors throughout Dane County, creating a specialized, highly lucrative Health IT cluster.
| Decade |
Key Milestone in Madison Healthcare IT Development |
| 1970s |
1979: Judy Faulkner founds Human Services Computing (Epic) in a Madison basement. |
| 1980s |
Mid-1980s: Company renamed to Epic. 1987: Release of Resolute Professional Billing. |
| 1990s |
1992: Launch of EpicCare, the first Windows-based EMR. 1997: Release of EpicWeb. |
| 2000s |
2000: Debut of MyChart patient portal. 2003: $1.8 billion Kaiser Permanente contract. |
| 2020s |
2024/2025: Massive investments in AI architecture, ambient listening, and predictive analytics. |
R&D Tax Credit Application and Eligibility
Software development presents some of the most complex and heavily audited challenges under federal and state R&D tax credit laws. The tax code recognizes that not all coding is created equal; routine software maintenance or building standard web applications does not meet the statutory requirements.
Qualifying Activities in Health IT:
- Algorithmic Optimization and AI Integration: Developing novel, highly complex algorithms for ambient listening and artificial intelligence (AI) charting capable of securely parsing millions of unstructured medical notes in real-time.
- Interoperability Systems Architecture: Designing the underlying backend architecture required to seamlessly integrate disparate hospital databases (radiology, pharmacy, billing) into a single, high-speed, secure, HIPAA-compliant patient portal.
- Cloud Infrastructure and Testing: Under recent IRS and Wisconsin DOR guidance, the costs associated with cloud computing time-sharing and hosting for dedicated development servers—environments where software engineers program, compile, and beta test new functionality—qualify as eligible supply expenses.
Case Law and Administrative Guidance: A critical juncture in healthcare IT tax compliance involves the legal classification of the software being developed. If software is developed to be sold, leased, or licensed to third parties (such as a Madison firm licensing software to a hospital system), it falls under the standard Four-Part Test. However, if a healthcare organization develops software exclusively for its own administrative or general management operations (e.g., internal HR tracking, proprietary financial ledgers, or standard inventory management), it triggers the highly stringent “Internal Use Software” (IUS) rules.
Under Proposed Treasury Regulation § 1.41-4(c)(6)(vi) and IRS Treasury Decision (TD) 9786, Internal Use Software must pass the standard Four-Part Test plus an additional three-part “High Threshold of Innovation” (HTI) test. The HTI test demands that the software:
- Innovation: The software must result in a reduction in cost or an improvement in speed or performance that is substantial and economically significant.
- Significant Economic Risk: The taxpayer must commit substantial financial resources to the software’s development with a high degree of uncertainty regarding the technical capability to successfully recover those resources.
- Commercial Unavailability: The software cannot be commercially available as an off-the-shelf solution for use by the taxpayer without undergoing massive, fundamental architectural modifications.
Firms within the Madison Health IT cluster must maintain rigorous contemporaneous documentation separating commercial-facing software engineering from internal administrative code. Because modern software architecture relies heavily on Agile development methodologies, the IRS expects taxpayers to produce detailed Jira tickets, system architectural schematics, pull-request histories, and code-commit logs to definitively prove that iterative software sprints constitute a genuine “process of experimentation”. Wisconsin DOR follows these exact IRS guidelines when assessing the state-level 5.75% software development claims, routinely rejecting claims that rely on vague, post-hoc employee interviews.
Case Study 3: Medical Imaging and Diagnostics
Industry Evolution in Madison
The medical imaging and diagnostics industry in Madison provides a textbook, historical example of how basic academic research translates directly into regional industrial dominance. The genesis of this cluster traces back to 1958, when UW-Madison physicist John R. Cameron initiated a collaborative program linking the physics department with nuclear medicine and radiology. Securing grants from the Atomic Energy Commission, Cameron attracted top-tier graduate talent, formally establishing the Medical Physics program. By 1981, this initiative had formalized into a robust, world-renowned academic department that cross-pollinated human oncology, physics, and nuclear engineering.
A pivotal, world-changing breakthrough occurred in the 1970s under the direction of Professor Charles Mistretta. Mistretta and his research team developed Digital Subtraction Angiography (DSA). By mathematically subtracting a baseline, pre-injection X-ray from a real-time image taken with an opaque tracer in the bloodstream, DSA allowed physicians to view real-time, unobstructed blood flow without the interference of background bones or soft tissues. This innovation dramatically reduced the toxicity of dye required and the radiation dose administered to patients, essentially creating the modern medical specialty of interventional radiology. Concurrently, UW-Madison medical physics professor Richard Mazess invented dual-energy x-ray absorptiometry (DXA) scanners to accurately detect osteoporosis and measure bone mineral density. Mazess spun this technology out of the university, founding the Madison-based Lunar Corporation in 1973.
General Electric (now GE HealthCare), which had been operating in the imaging space since acquiring Victor Electric in 1920, recognized the unparalleled density of imaging talent in Madison. In 2000, GE Healthcare acquired Lunar Corporation to consolidate its position in bone densitometry. Today, GE HealthCare (which spun off as an independent public company in 2023) continues to operate massive, sophisticated R&D and manufacturing facilities within Wisconsin. These corporate entities maintain a continuous, highly productive feedback loop with UW-Madison researchers, allowing for the rapid optimization of MRI clinical applications, Deep Learning Image Reconstruction (DLIR) neural networks, and computed tomography (CT) protocols before commercialization.
| Timeline of Medical Imaging Development |
Major Milestones |
| 1958 |
John R. Cameron establishes the Medical Physics program at UW-Madison. |
| 1970s |
Charles Mistretta develops Digital Subtraction Angiography (DSA); Richard Mazess founds Lunar Corporation for DXA scanners. |
| 1980s-1990s |
UW-Madison develops advanced MRI techniques (3D TRICKS, VIPR). GE expands through acquisitions (Resonex, Marquette Medical). |
| 2000 |
GE Healthcare acquires Lunar Corporation, cementing Madison as a core hardware and software R&D hub. |
| 2023 |
GE HealthCare spins off into an independent publicly traded company, continuing massive R&D investments. |
R&D Tax Credit Application and Eligibility
The medical imaging sector operates at the bleeding edge of both high-level software engineering and complex, heavy hardware fabrication, opening multiple, massive avenues for R&D tax credit claims at both the federal and state levels.
Qualifying Activities in Medical Imaging:
- Hardware Prototyping and Fabrication: Designing, fabricating, and meticulously assembling experimental pilot models of new magnetic resonance imaging (MRI) coils, superconducting magnets, or digital PET/CT scanner components.
- Algorithm and Software Development: Engineering highly complex neural networks for prescriptive and predictive analytics, such as machine learning systems that can computationally reduce X-ray radiation doses while simultaneously maintaining high-resolution diagnostic image quality.
- Regulatory Testing: Conducting exhaustive non-destructive testing, radiation shielding evaluations, and clinical safety studies required to secure rigorous U.S. Food and Drug Administration (FDA) clearance for new medical devices.
Case Law and Administrative Guidance: One of the most legally complex areas of R&D tax law involves the treatment of prototype and pilot model manufacturing costs. Medical imaging equipment is exceptionally expensive to design and fabricate. In the recent, highly influential Tax Court case Intermountain Electronics, Inc. v. Commissioner (2024), the court evaluated whether the heavy production expenses incurred in developing a physical pilot model qualified as a legitimate process of experimentation under IRC Section 41(d)(3)(A).
The Tax Court confirmed that costs for both raw supplies and the wages of non-production and production staff involved in fabricating and testing an experimental pilot model—prior to the design being fully validated and finalized for commercial mass production—can absolutely qualify as eligible expenditures under Section 174 and Section 41. For Madison’s medical device manufacturers, this precedent underscores the absolute necessity of clearly documenting the exact chronological point at which a physical machine transitions from an experimental, uncertain pilot model (eligible for the credit) to a finalized, commercially viable asset (ineligible for the credit).
Additionally, large multinational corporate entities operating in this space often utilize the IRS ASC 730 Directive. The IRS allows large corporate taxpayers who expense their R&D costs on their Certified Audited Financial Statements under U.S. Generally Accepted Accounting Principles (GAAP ASC 730) to follow specific administrative safe harbors for computing their QREs. This directive significantly streamlines the grueling substantiation process for massive, multi-departmental imaging research budgets, reducing the burden of traditional IRS audits while ensuring compliance.
Case Study 4: Nuclear Engineering and Fusion Technology
Industry Evolution in Madison
While nuclear fusion has traditionally been viewed by the public as a monolithic, multi-decade quest for limitless grid energy, a highly specialized, pragmatic commercial cluster has emerged in the Madison area focusing on near-term industrial applications of fusion technology. This sector relies heavily on UW-Madison’s historic strength in nuclear engineering, a program that has operated continuously for over 50 years and holds the prestige of producing the very first U.S. PhD in fusion research.
The commercialization of this local expertise began in earnest in 2005 when UW-Madison graduate Dr. Greg Piefer founded Phoenix Nuclear Labs. The company’s foundational premise was that the fastest, most economically viable path to achieving eventual fusion energy was to commercialize near-term industrial applications of the technology, thereby allowing for iterative improvement and continuous financial reinvestment. Phoenix initially pioneered fusion-based technology to generate neutrons for industrial non-destructive testing and radiation hardening.
Recognizing the immense potential in the medical field, Piefer spun out SHINE Technologies in 2010. Operating out of massive facilities in nearby Janesville (firmly within the Madison technological and labor orbit), SHINE applies subcritical nuclear fusion and highly advanced radiochemical separation techniques to produce critical medical isotopes. Specifically, they focus on producing Lutetium-177 (Lu-177), a highly sought-after isotope utilized in advanced, targeted cancer therapies. By 2021, SHINE re-acquired Phoenix to consolidate their fusion technological advantage under a single corporate umbrella, creating a vertically integrated nuclear technology powerhouse heavily staffed by UW-Madison alumni.
R&D Tax Credit Application and Eligibility
The fusion and medical isotope manufacturing industry incurs massive capital expenditures, highly specialized engineering personnel costs, and extreme regulatory oversight, generating massive pools of Qualified Research Expenses.
Qualifying Activities in Nuclear Engineering:
- Reactor Design and Particle Accelerator Architecture: Engineering the physical, magnetic, and thermodynamic architecture of subcritical accelerator-based facilities required to safely generate Cherenkov radiation and efficiently produce medical isotopes.
- Regulatory Compliance Engineering: Developing complex mathematical models, fluid dynamic simulations, and structural integrity stress tests required to secure landmark construction permits and operating approvals from the rigorous Nuclear Regulatory Commission (NRC).
- Transmutation and Radiochemical Processes: Experimenting with novel radiochemical separation techniques to rapidly isolate highly pure medical isotopes from irradiated target materials without causing dangerous environmental contamination.
Case Law and Administrative Guidance: Entities operating in the nuclear, aerospace, and defense sectors frequently receive substantial grants, subsidies, or contracts from federal entities such as the Department of Energy (DOE) or the Department of Defense (DOD). Consequently, they face intense, highly specialized IRS scrutiny regarding the “Funded Research Exclusion” outlined under IRC Section 41(d)(4)(H).
The seminal case Fairchild Industries, Inc. v. United States established the modern legal framework for analyzing this exclusion. The Federal Circuit Court of Appeals ruled that if a taxpayer’s payment from a government entity or a prime contractor is explicitly contingent on the ultimate success of the research, the taxpayer bears the financial risk of the endeavor, and the research is not considered funded (thus rendering the QREs eligible for the credit). Conversely, if the government pays the taxpayer a fixed sum to merely attempt the research regardless of the final scientific outcome, the government bears the risk, and the taxpayer is legally barred from claiming the credit.
Furthermore, as elaborated in Lockheed Martin Corp. v. United States and recently reaffirmed in the Smith v. Commissioner architecture case, bearing the financial risk is only half the battle; the taxpayer must also legally retain “substantial rights” to the research results. If a Madison-based fusion company develops a revolutionary new neutron-generating technique under a federal grant, but the contract explicitly grants the government exclusive ownership of the intellectual property, prohibiting the company from using that IP for commercial medical isotope production, the company has not retained substantial rights. In such a scenario, the QREs are entirely disqualified. Thus, meticulous contract drafting, legal review, and IP management are paramount for financial survival in this sector.
State-Level Nuances: The Energy Efficiency Enhanced Credit
For advanced engineering firms in Wisconsin, the state provides a powerful, highly specialized secondary tax incentive that surpasses the federal framework. Under Wis. Stat. § 71.07(4k)(a) and § 71.28(4)(ab)2, research activities directed toward “certain energy efficient products” or the design of “internal combustion engines” qualify for an enhanced 11.5% R&D tax credit, exactly double the state’s general 5.75% rate.
Crucially, the Wisconsin statutory definition of an “internal combustion engine” is remarkably broad and forward-looking; it explicitly includes substitute modern products such as fuel cell, electric, and hybrid drive systems used in vehicles, generators, and industrial machinery. Additionally, research into energy-efficient lighting systems, building automation, and advanced automotive batteries qualifies for this 11.5% tier.
If a Madison-area engineering firm conducts research into advancing fuel cell thermodynamic efficiency, modifying electric hybrid drive trains for heavy construction equipment, or developing next-generation battery storage systems, these specific QREs must be strictly segregated in the company’s project accounting systems. The Wisconsin Tax Appeals Commission enforces strict nexus requirements for this enhanced credit; if a company cannot unequivocally tie the specific engineering labor hours and supply costs directly to the hybrid or energy-efficient component, the Department of Revenue will aggressively reclassify the expenses down to the lower 5.75% tier, or disallow them entirely during an audit.
Case Study 5: Food Science, Dairy Innovation, and Agricultural Technology
Industry Evolution in Madison
Agriculture and food science form the historical and economic bedrock of Wisconsin. In the mid-19th century, Wisconsin was the nation’s primary producer of wheat, considered “America’s breadbasket”. However, severe soil nutrient depletion, compounded by devastating chinch bug infestations in the 1860s, threatened complete agricultural collapse and forced a catastrophic economic pivot. Supported heavily by the early agricultural research departments at UW-Madison, the state transitioned aggressively from fragile wheat cash crops to resilient dairy farming.
In 1887, UW-Madison hired chemist Stephen Babcock, who invented the first reliable, standardized test for measuring the butterfat content of milk, fundamentally stabilizing and commercializing the modern dairy industry. Shortly thereafter, foundational nutritional experiments conducted at the university by E.B. Hart and E.V. McCollum led directly to the scientific discovery of Vitamins A and B, changing global understandings of human and animal diets. This unparalleled legacy of food science is currently institutionalized within the Center for Dairy Research (CDR) at UW-Madison, a world-class facility that partners closely with the Dairy Business Innovation Alliance. The CDR provides direct technical assistance, grants, and commercialization strategies for food processors and cheese makers across the state. Today, Madison’s food and beverage cluster encompasses over 3,700 companies statewide, seamlessly integrating complex biotechnology into modern food production, shelf-life stabilization, and hybrid crop development.
R&D Tax Credit Application and Eligibility
The food, beverage, and broader agricultural industry frequently overlooks the massive financial potential of the R&D tax credit, operating under the widespread misconception that the incentive is strictly reserved for individuals wearing lab coats in software development or pharmaceutical laboratories. However, modern food science relies heavily on the hard scientific principles of chemistry, biology, and thermodynamics, perfectly satisfying the “Technological in Nature” statutory test.
Qualifying Activities in Food Science:
- Product Formulation and Recipe Engineering: Developing entirely new formulations to achieve a highly specific nutritional profile (e.g., engineering a high-protein, low-sodium cheese) without compromising structural integrity, melting points, or consumer taste profiles.
- Process Optimization and Manufacturing: Engineering novel pasteurization, fermentation, or milling techniques to increase automated production throughput, reduce biological waste, or extend the ambient shelf-life of perishable goods.
- Scale-Up Operations: Translating a successful small-scale test-kitchen formulation to massive, industrial mass-production machinery. This inherently involves severe technical uncertainties regarding thermodynamics, fluid viscosity under pressure, and the mechanical shear forces applied to the food matrix.
Case Law and Administrative Guidance: While agricultural activities can qualify, this sector faces severe, unique hurdles regarding the “Process of Experimentation” test, primarily due to historically poor documentation practices within the industry. The defining, highly consequential judicial precedent in this sector is the Tax Court case Siemer Milling Company v. Commissioner of Internal Revenue (2019).
Siemer Milling, a family-owned wheat flour producer, claimed substantial research credits for developing new, innovative products such as wheat hybrids and heat-treated flour. The IRS audited the claims and denied the credits entirely, and the U.S. Tax Court upheld the IRS’s denial. Crucially, the court’s rationale was not that food science fails to qualify as research; rather, the taxpayer catastrophically failed to provide adequate documentation proving they engaged in a “methodical plan involving a series of trials to test a hypothesis, analyze the data, refine the hypothesis, and retest the hypothesis so that it constitutes experimentation in the scientific sense”.
The taxpayer presented generalized, after-the-fact summaries of their product development efforts, which the court aggressively dismissed as mere “conclusory statements” lacking the rigorous, contemporaneous documentation required by law. Siemer Milling serves as a critical, binding warning for Madison’s food science and dairy innovators. While discovering a new fermentation process or developing a hybrid crop undeniably relies on biology and chemistry, the company must meticulously document the scientific method in real-time. This includes recording the baseline hypothesis, the exact variables adjusted during test batches, the specific chemical or physical results of each test, and the subsequent formulation changes.
Fortunately for taxpayers, the court in Siemer did issue a taxpayer-friendly ruling on one specific point: it explicitly established that technical uncertainties need not be solved within a single tax year. The court acknowledged that complex agricultural innovations often span multiple periods, allowing valid research expenses to be claimed across several tax years even if the ultimate solution remains elusive. Furthermore, as reinforced in George v. Commissioner, agricultural entities must ensure that the personnel conducting these tests possess actual scientific backgrounds, rather than merely claiming the wages of standard farmhands or line workers.
Tax Administration Guidance, Audit Defense, and Documentation Strategies
Filing for the United States federal and Wisconsin state R&D tax credits requires navigating a complex labyrinth of administrative guidance. The financial rewards are substantial, but they are matched by the intensity of regulatory scrutiny. Taxpayers must implement proactive risk mitigation and documentation strategies to survive audits.
The Federal and State Audit Environment
At the federal level, the IRS relies heavily on its published “Audit Techniques Guide (ATG) for Credit for Increasing Research Activities”. The ATG explicitly directs field examiners to heavily scrutinize oral testimonies and post-hoc estimates. R&D tax credit studies generated purely by interviewing personnel months or years after the research was completed are frequently rejected. The IRS demands contemporaneous documentation—real-time tracking systems mapped directly to specific business components, laboratory notebooks, CAD architectural drawings, and prototype testing logs. Recently, the IRS has implemented a rigorous “Classifier review system,” as highlighted in the 2024 case Meyer, Borgman & Johnson, Inc. v. Commissioner, which can unilaterally deny poorly substantiated refund claims before they even reach a formal field examiner.
At the state level, the Wisconsin Department of Revenue utilizes its own comprehensive manual, Publication 131, to guide taxpayers and auditors. In practice, the DOR generally follows IRS determinations; if the IRS formally audits a Madison-based company and approves specific QREs, the DOR will typically adopt that finding without duplicating the audit. However, this deference has limits. If the IRS audit scope excluded Wisconsin-specific operations, or if the taxpayer is aggressively claiming the state’s enhanced 11.5% engine/energy credit, the DOR conducts independent, highly detailed audits.
The Wisconsin Tax Appeals Commission strictly enforces the requirement that costs must be captured and documented at the specific “business component” level to establish a direct, undeniable nexus between the financial expense and the qualified scientific activity. Generic cost-center accounting—such as simply claiming all W-2 wages from the “Engineering Department” without tying them to specific projects—is routinely rejected by the state in favor of rigorous, project-based accounting.
Summary of Foundational Case Law
The interpretation and enforcement of both federal and Wisconsin R&D tax credit statutes rely heavily on the following established judicial precedents. All Madison-based enterprises must incorporate the lessons from these rulings into their corporate compliance frameworks to ensure their credits survive regulatory scrutiny:
| Legal Precedent |
Industry Context |
Judicial Finding / Principle Established |
| Siemer Milling Co. v. Comm’r |
Food Science / Agriculture |
Disallowed credits entirely due to a severe lack of contemporaneous documentation proving a methodical process of experimentation. Affirmed that technical uncertainty can legally span multiple tax years. |
| Fairchild Industries v. United States |
Aerospace / Engineering |
Established the definitive rule that research is not “funded” (and thus qualifies for the credit) if the taxpayer bears the financial risk of failure, regardless of the existence of government contracts. |
| Smith v. Comm’r |
Architecture / Engineering |
Denied IRS summary judgment on funded research claims, emphasizing the legal necessity of analyzing specific contractual clauses regarding the retention of “substantial rights” to the resulting intellectual property. |
| Intermountain Electronics v. Comm’r |
Manufacturing / Electronics |
Affirmed that both non-production and production costs incurred to develop, fabricate, and test experimental pilot models can qualify as eligible QREs prior to commercialization. |
| George v. Comm’r |
Agriculture / Biology |
Disallowed significant portions of agricultural claims that lacked personnel capable of conducting hard-science research, reaffirming the strict, uncompromising nature of the “Technological in Nature” test. |
| Meyer, Borgman & Johnson v. Comm’r |
General Engineering |
Demonstrated that the IRS Classifier review system will deny refund claims outright if they lack a clear narrative explaining the process of experimentation and detailed breakdowns of business components. |
Final Thoughts
The United States federal and Wisconsin state Research and Development tax credits represent highly lucrative, yet administratively complex, fiscal incentives designed to reward the financial risks inherent in technological innovation. Madison, Wisconsin, powered by the profound historical legacy and ongoing multi-billion-dollar investments of the University of Wisconsin-Madison and the Wisconsin Alumni Research Foundation, has cultivated a dense, world-class ecosystem of high-technology industries perfectly positioned to capitalize on these legal frameworks.
From the biochemical breakthroughs of the biohealth and food science sectors, to the computational architecture of healthcare information technology, and the heavy, capital-intensive engineering of medical imaging and nuclear fusion, Madison’s enterprises continuously push the boundaries of global technological advancement. However, as demonstrated by the relentless evolution of federal legislative code, administrative regulations, and strict judicial scrutiny, achieving a scientific breakthrough is only half the battle.
To successfully and legally monetize these innovations via the tax code, organizations must implement sophisticated, real-time project accounting systems. They must unequivocally substantiate the rigorous Four-Part Test with contemporaneous documentation, meticulously navigate the perilous contractual boundaries of the funded research exclusion, and accurately segregate costs to capture Wisconsin’s specialized 11.5% enhanced energy and engineering incentives. By aligning aggressive innovation with strict statutory compliance, Madison’s enterprises can secure the capital necessary to maintain their position at the forefront of global research and development.
The information in this study is current as of the date of publication, and is provided for information purposes only. Although we do our absolute best in our attempts to avoid errors, we cannot guarantee that errors are not present in this study. Please contact a Swanson Reed member of staff, or seek independent legal advice to further understand how this information applies to your circumstances.














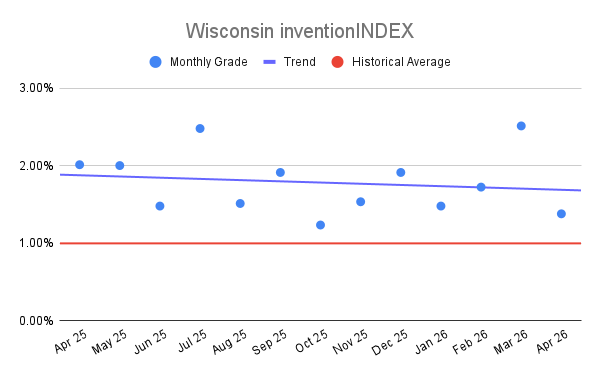 Wisconsin inventionINDEX April 2026:[...]
Wisconsin inventionINDEX April 2026:[...] 
